Mgco3 + Hcl Balanced Equation With States Drawi
Al2O3+HCl=AlCl3+H2O balance the chemical equation. Aluminium oxide and hydrogen chloride reaction My documentary 6.61K subscribers Subscribe 1 2 3 4 5 6 7 8 9 0 1 2 3 4 5 6 7 8 9 0 1 2 3 4.

SOLVEDAn electrolytic cell produces Al and O2 from Al2O3 at a rate of 5.00 kg per day (1.00 day
The HCl and Al2O3 reaction can be balanced by the hit and trial method according to the steps given below - HCl (aq) + Al2O3(s)= AlCl3(aq)+ H2O (l) As the reactant contains two aluminium atoms so two moles of the aluminium chloride must be present on the reactant side in order to balance the reaction. HCl (aq) + Al2O3(s)= 2AlCl3(aq)+ H2O (l)

How to balance NaOH + HCL Balanced chemical equationStepbystep explained YouTube
Al2O3 + 6 HCl → 2 AlCl3 + 3 H2O - Balanced equation | Chemical Equations online! Solved and balanced chemical equation Al2O3 + 6 HCl → 2 AlCl3 + 3 H2O with completed products. Application for completing products and balancing equations.

How to Write the Net Ionic Equation for Al2O3 + HCl Note Al2O3 should be (s). YouTube
1. F e+Al2O3→ 2. Cu+M gCl2 → 3. Cu+AlCl3→ 4. F e+HCl→ Q. Among Al2O3, SiO2, P 2O3, SO2 the correct order of acid strength is Al2O3 + HCl vrightarrow text

Na2CO3+HCl=NaCl+CO2+H2O Balanced EquationSodium carbonate+Hydrochloric acid=Sodium chloride
Let's balance this equation using the inspection method. First, we set all coefficients to 1: 1 Al 2 O 3 + 1 HCl = 1 H 2 O + 1 AlCl 3 For each element, we check if the number of atoms is balanced on both sides of the equation. Al is not balanced: 2 atoms in reagents and 1 atom in products. In order to balance Al on both sides we: Multiply.

Mgco3+Hcl=Mgcl2+H2O+Co2 Ionic Equation delantalesybanderines
Al2O3 | aluminium oxide | solid + HCl | hydrogen chloride | solid = AlCl3 | aluminium chloride | solid + H2O | water | solid | Temperature: temperature, Other Condition excess chlorine Introduce Detailed information about the equation Reaction conditions when applied Al2O3 + HCl Reaction process Al2O3 + HCl The result of the reaction Al2O3 + HCl

54.0g Al reacts with 64.0g O2 to form Al2O3 according to the equation. 4Al+3O2 = 2Al2O3 O2 32 g
HCl + Al2O3 = AlCl3 + H2O is a Double Displacement (Metathesis) reaction where six moles of Hydrogen Chloride [HCl] and one mole of Aluminum Oxide [Al 2 O 3] react to form two moles of Aluminum Chloride [AlCl 3] and three moles of Water [H 2 O] Show Chemical Structure Image Reaction Type Double Displacement (Metathesis) Exchange reaction Reactants

How to balance Al2O3+C=Al+CO2Chemical equation Al2O3+C=Al+CO2Al2O3+C=Al+CO2 balancedAl2O3+C
Balanced Chemical Equation - Solution. Al 2 O 3 + 6 HCl → 2 AlCl 3 + 3 H 2 O. The coefficients show the number of particles (atoms or molecules), and the indices show the number of atoms that make up the molecule. New substances are formed as a result of the rearrangement of the original atoms.

Find the mass of HCl that must react when starting with 23.2 grams of Al2O3, according
HCl + Al2O3 = AlCl3 + H2O - Chemical Equation Balancer Balance the reaction of HCl + Al2O3 = AlCl3 + H2O using this chemical equation balancer! ChemicalAid ChemicalAid ZeptoMath Elements Periodic Table Periodic Trends Element Charts Future Elements Calculators Chemical Equation Balancer Empirical Formula Calculator Molar Mass Calculator

Aluminum oxide (Al2O3) and iron (Fe) react according to the following equation _Al2O3 + _Fe →
Balanced Chemical Equation 2AlCl3·6H2O → Al2O3 + 6HCl + 9H2O ⬇ Scroll down to see reaction info and a step-by-step answer, or balance another equation. Reaction Information Word Equation Aluminum Chloride Hexahydrate = Aluminum Oxide + Hydrogen Chloride + Water

Ideal Naoh Hcl Balanced Equation What Is The Chemical For Photosynthesis
Chem Exam 3. Get a hint. Find the mass of AlCl3 that is produced when 25.0 grams of Al2O3 react with HCl according to the following balanced equation. Al2O3 (s) + 6HCl (aq) → 2AlCl3 (aq) + 3H2O (l) 72.9 g. 16.3 g.
PPT 30 Nov. 2010 Law of Conservation of Mass PowerPoint Presentation ID1532140
Reactants Aluminium - Al Aluminum 13Al Element 13 Al Molar Mass Al Oxidation Number Ammonium Perchlorate - NH 4 ClO 4 Ap NH4ClO4 Molar Mass NH4ClO4 Oxidation Number Products Aluminum Oxide - Al 2 O 3 Aluminium Oxide Poraminar Martoxin Lucalox Ketjen B Jubenon R Hypalox Ii Fasertonerde Al2O3 Molar Mass Al2O3 Oxidation Number Hydrogen Chloride - HCl

SOLVED Given the following thermochemical equations 4Al(s) 4Al(s) 301(g) 2Al,O3(s) 3MnOz(s
Balanced Chemical Equation Al 2 O 3 + 6 HCl → 2 AlCl 3 + 3 H 2 O ⬇ Scroll down to see reaction info and a step-by-step answer, or balance another equation. Reaction Information Word Equation Aluminum Oxide + Hydrogen Chloride = Aluminum Chloride + Water

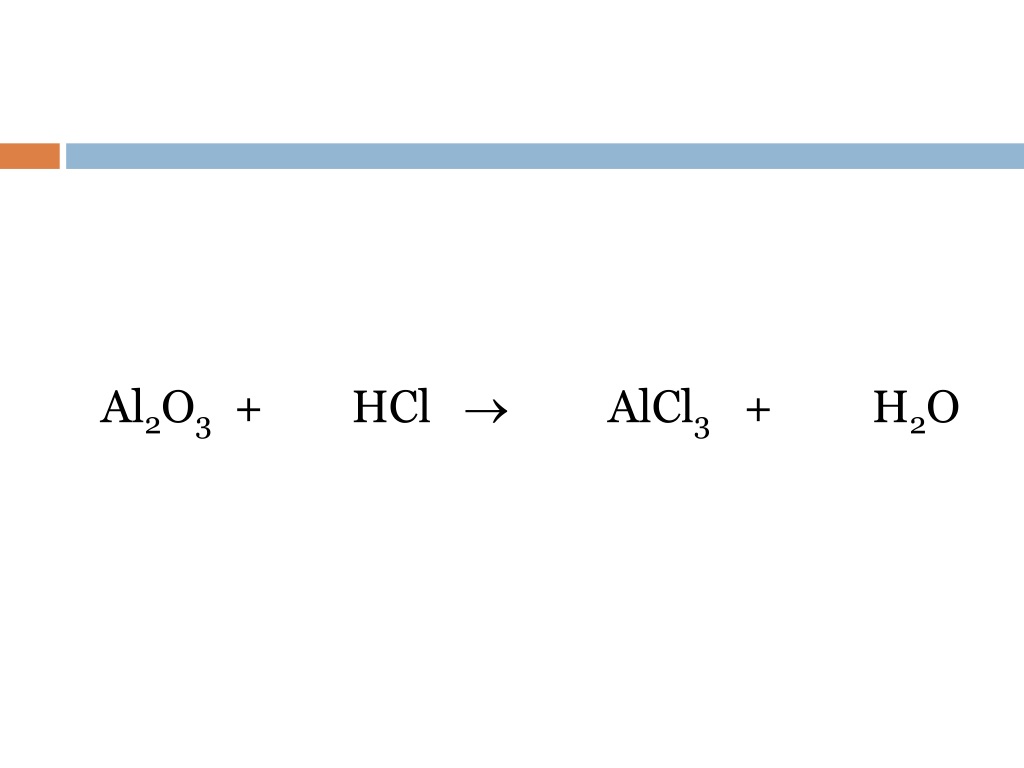
Balance this equation Al2O3+ HCl AlCl3 + H2O Science Metals and Nonmetals 12348429
Balance the oxygen atoms by placing a coefficient of 2 in front of H 2 O: H 2 + O 2 = 2H 2 O. Now, there are 4 H atoms on the right side, so we adjust the left side to match: 2H 2 + O 2 = 2H 2 O. Check the balance. Now, both sides have 4 H atoms and 2 O atoms. The equation is balanced.

NiFoamStructured NiAl2O3 Ensemble as an Efficient Catalyst for GasPhase Acetone
Let's balance this equation using the inspection method. First, we set all coefficients to 1: 1 Al 2 O 3 + 1 HCl = 1 Al 2 Cl 6 + 1 H 2 O For each element, we check if the number of atoms is balanced on both sides of the equation. Al is balanced: 2 atoms in reagents and 2 atoms in products. O is not balanced: 3 atoms in reagents and 1 atom in.

Al + hcl balanced equation
How to Balance Al2O3 + HCl = AlCl3 + H2O (Aluminum Oxide + Hydrochloric Acid) Wayne Breslyn 727K subscribers Join Subscribe Subscribed 812 75K views 5 years ago In this video we'll balance.